GI Microbiome Panel
Understanding the state of the microbiome in the gut can help you manage your GI cases and provide better care.
Microbiome Testing – Overview
Canine or Feline
The gut microbiome is at the center of gastrointestinal, immune, and metabolic health. VDI’s GI Microbiome Panel provides veterinarians with a comprehensive analysis of the intestinal ecosystem — identifying microbial imbalance, loss of diversity, and functional disruption that underlie many chronic GI and systemic conditions. Unlike routine fecal testing, which detects only pathogens, this panel measures the full microbial community using advanced molecular profiling and integrates results into the Dysbiosis Severity Index (DSI) for clear clinical interpretation. By quantifying the degree and impact of dysbiosis, veterinarians can move beyond symptomatic treatment to target the root cause, personalize therapy, and track recovery over time.
From imbalance to insight — the GI Microbiome Panel transforms complex data into actionable diagnostics for every GI case.

QUICK INFO
| Test Methodology: | Next-Gen DNA Sequencing (NGS) |
| Species: | Canine or Feline |
| Sample Type: | Stool (fecal) – fresh (requires microbiome collection kit) |
| Fasting: | Not Required |
| GI Microbiome Panel* | Culture Testing | PCR Testing | |
|---|---|---|---|
| Diagnosis of ALL bacterial and fungal pathogens | YES | Only those that can grow in a lab | Limited to target panels |
| Antibiotic Resistance Profiling | YES | Only those that can grow in a lab | Limited to target panels |
| Pathogen Quantification | YES | Only those that can grow in a lab | Limited to target panels |
| Incubation/Growth Independent | YES | NO | YES |
| Ambient Shipping/Storage Temp | YES | NO | NO |
| Microbiome Profiling | YES | NO | YES |
What’s included?
- Complete bacteria and fungal quantification
- List of potential clinically relevant species
- Antibiotic resistance for relevant species, including a table of common antibiotics upon request
- Diversity Severity Index (DSI)
- Richness, Evenness, Fermenter scoring
- Recommended actions based on findings
- Dietary recommendations
- Product recommendations
Sample Collection
The GI Microbiome Panel requires a stool sample collected and submitted using a Microbiome Collection Kit (MCK). Available through VDI.
Learn more about VDI Microbiome Testing
Table of Contents – click a section to jump to it.
Section 1: Dysbiosis Severity Index (DSI)
Section 2: Why Microbial Indices Matter
Section 3: Toxins and the Microbiome
Section 4: Vitamin D & B12: Hidden drivers of Microbiome Health
Dysbiosis Severity Index (DSI)
Measuring the Microbial Imbalance That Drives GI Disease
Overview
Dysbiosis refers to a disruption in the normal balance of the intestinal microbiota—where beneficial, commensal organisms decline and opportunistic or pathogenic species expand. In dogs and cats, this imbalance can impair short-chain fatty acid production, alter bile acid metabolism, weaken the intestinal barrier, and drive chronic mucosal inflammation. These microbial shifts are often central to the persistence of gastrointestinal signs such as diarrhea, vomiting, bloating, and malabsorption, even when routine lab tests appear normal.
VDI’s Dysbiosis Severity Index (DSI) provides veterinarians with a quantitative measure of this imbalance. Unlike conventional dysbiosis scores that simply indicate the presence or absence of disruption, the DSI goes further—it ranks the severity and functional impact of the dysbiosis on the patient’s overall gastrointestinal health. The DSI integrates multiple microbial, metabolic, and clinical parameters to deliver a clear, tiered assessment that guides diagnosis, treatment selection, and monitoring of response over time.
By quantifying both the degree and biological consequence of microbial imbalance, the Dysbiosis Severity Index transforms the interpretation of GI microbiome data from descriptive to actionable—empowering veterinarians to personalize care, track progress, and restore balance where it matters most.
When to Test for Dysbiosis
The Dysbiosis Severity Index (DSI) is most valuable when gastrointestinal signs are present but standard diagnostics (CBC, chemistry, imaging) fail to reveal a clear cause. Testing is recommended for:
- Chronic or recurrent diarrhea, vomiting, or flatulence
- Suspected inflammatory bowel disease (IBD) or chronic enteropathy
- Antibiotic- or diet-responsive GI conditions
- Unexplained weight loss, poor appetite, or malabsorption
- Cases of suspected microbiome disruption following antibiotics, steroids, or toxins
By establishing a baseline DSI, veterinarians can monitor recovery and evaluate treatment response over time. Serial testing documents whether the microbiome is normalizing or remains impaired, helping to refine diet, probiotic, or antimicrobial strategies.
Key Features of the Dysbiosis Severity Index
- Exclusive to VDI Laboratory – developed from proprietary datasets in companion animals
- Quantifies both imbalance and impact – measures not only presence of dysbiosis but its biological severity
- Integrates multi-parametric data – combines multiple measured indicators into one comprehensive index
- Tracks progress over time – ideal for monitoring therapy effectiveness and relapse risk
Interpreting the DSI
The Dysbiosis Severity Index (DSI) expresses the degree of microbiome imbalance on a continuous, quantitative scale—translating complex microbial and metabolic data into a clear clinical context. The index not only identifies the presence of dysbiosis but also reflects its biological impact on the patient’s gastrointestinal function.
| DSI Category | Interpretation | Clinical Implication |
| Normal | Balanced microbiome. Stable core commensal population and normal metabolite profiles. | No significant microbiome disruption; monitor as baseline. |
| Mild | Early or transient imbalance; often seen post-antibiotic, diet change, or stress. | Often self-limiting; address underlying cause, consider dietary or probiotic support. |
| Moderate | Functionally significant dysbiosis impacting gut homeostasis. | Indicates ongoing disruption—treat underlying condition and support microbiome recovery. |
| High | Severe dysbiosis with functional compromise and inflammatory risk. | Strongly associated with GI disease, poor nutrient absorption, or chronic inflammation; requires targeted intervention and follow-up testing. |
How to use the DSI in Practice
The Dysbiosis Severity Index represents the next generation of microbiome diagnostics—transforming raw sequencing data into an actionable clinical tool that helps veterinarians move from observation to intervention with confidence.
- Baseline assessment: Establish before or at the start of therapy to identify underlying dysbiosis.
- Monitoring tool: Repeat testing after dietary, probiotic, or antibiotic intervention to document recovery or relapse.
- Integrative insight: Combine with other VDI markers—such as vitamin B12, folate, magnesium, or inflammation biomarkers—for a comprehensive picture of gastrointestinal and metabolic health.
The GI Community
Imagine a cross-section of a thriving city, representing the gut ecosystem. Each tier occupies a distinct zone — from the busy street level (Core Commensals) to the hidden shadows (Fungi). All thriving cities need balance.
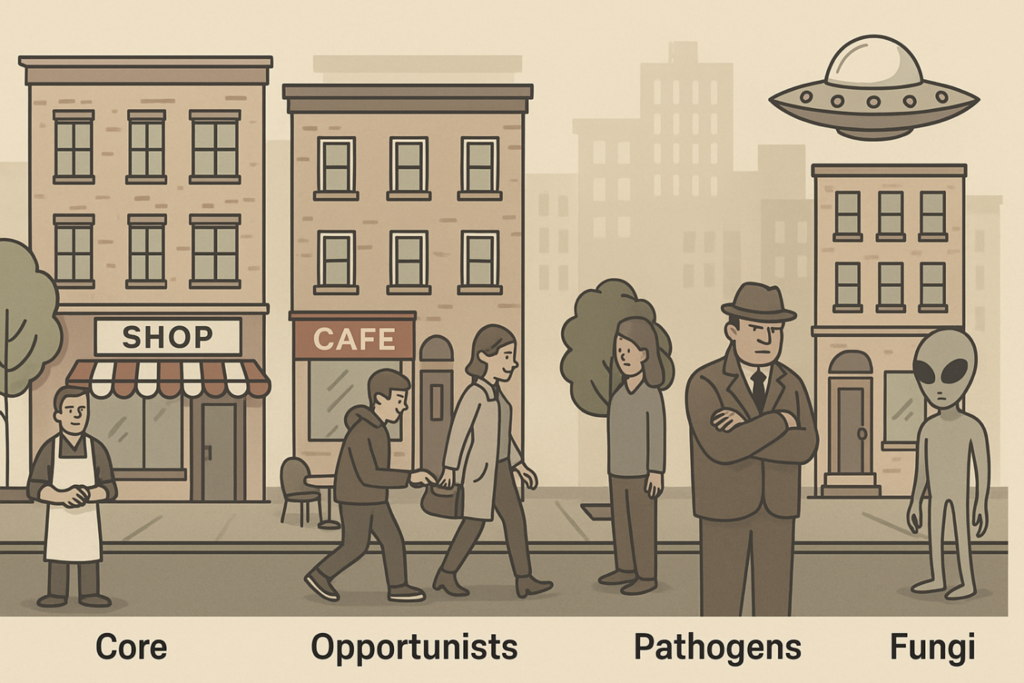
Tier 1: Core Commensals — The Shopkeepers
These are the reliable residents who keep the community running smoothly. Core commensals such as Faecalibacterium, Ruminococcus, Blautia, and Clostridium hiranonis produce short-chain fatty acids (SCFAs) that nourish intestinal cells, regulate pH, maintain barrier integrity, and modulate immune tolerance.
When this group is thriving, the gut is stable and resilient. When they decline—due to antibiotics, inflammation, or diet imbalance—the city loses its essential workers, setting the stage for instability and opportunistic activity.
Tier 2: Opportunistic Bloomers — The Pickpockets
Opportunists are normal residents that generally behave—until law and order break down. Think of them as pickpockets or petty criminals who take advantage of distraction. Examples include Enterococcus, Clostridium perfringens, and Escherichia/Shigella.
Under normal circumstances, the core commensals keep them in check through competition for nutrients and space. But when the core weakens, opportunists can bloom, stealing resources, producing gas, or triggering low-grade inflammation. Their overgrowth often marks mild to moderate dysbiosis—a signal that the gut ecosystem is stressed but still recoverable.
Tier 3: Pathogens — The Organized Crime Syndicate
These are the true disruptors. Pathogenic bacteria—such as Salmonella, Campylobacter, or toxigenic Clostridium difficile—move in when defenses fail. Like organized crime, they exploit chaos, damage infrastructure, and leave inflammation and toxicity in their wake.
Their presence indicates severe dysbiosis or active infection, requiring targeted therapy. Once they establish dominance, even beneficial bacteria struggle to regain territory, and recovery can be slow without focused intervention.
Tier 4: Fungi and Yeasts — The Aliens
Fungi are the outsiders—unseen most of the time, yet capable of dramatic disruption when conditions favor their rise. Species such as Candida or Cyniclomyces may bloom following antibiotic use or immune suppression. Their behavior is unpredictable, often producing metabolites or toxins that alter microbial dynamics and provoke immune confusion.
Like aliens visiting an otherwise familiar city, fungal blooms bring instability and uncertainty. Their presence usually signifies profound ecological imbalance or metabolic disturbance, and their control requires restoring bacterial dominance rather than simply eliminating fungi.
Why The Tiers Matter
Understanding these four tiers turns microbiome analysis from a list of organisms into a functional ecosystem assessment.
- Core commensals reflect stability and resilience.
- Opportunists signal imbalance.
- Pathogens denote breakdown and disease.
- Fungi warn of deeper systemic disruption.
Together, they provide a clear map of where the microbial community stands—and where it’s heading—allowing veterinarians to make informed, tier-based decisions in diagnosis, therapy, and monitoring.
Why Microbial Indices Matter
Beyond The Tiers
While the tiered classification describes who is living in the gut community, the supporting indices describe how healthy and functional that community truly is. Together, these quantitative measures provide veterinarians with a deeper view of microbial performance — revealing whether the ecosystem is robust, imbalanced, or collapsing under strain.
Total Bacterial Count —
The Population Census
The total bacterial count (TBC) measures the overall number of bacteria present in the fecal microbiome.
In a healthy gut, high total counts reflect a dense and active microbial population capable of nutrient breakdown, fermentation, and immune regulation.
A low total count often indicates microbial depletion — the “empty city” scenario — caused by antibiotics, poor diet, or inflammation, leaving room for opportunistic and pathogenic invaders.
Conversely, excessively high counts may occur with bacterial overgrowth or poor digestive clearance. TBC serves as the baseline context for interpreting all other indices: without knowing how populated the microbiome is, diversity and balance metrics lose meaning.
Evenness —
The Balance of Power
Evenness measures how equally the species present share the ecosystem. In a balanced microbiome, no single group dominates; resources and influence are evenly distributed, maintaining ecological stability. Loss of evenness — where a few taxa bloom while others collapse — signals dysbiosis in progress.
It’s the microbial equivalent of a city taken over by one industry or a few powerful groups, leading to inefficiency, loss of diversity, and vulnerability to disruption. Evenness is especially useful for detecting subtle imbalances before overt dysbiosis develops.
Richness —
The # of Different Residents
Richness represents how many unique microbial species inhabit the gut.
A rich microbiome is like a city with many different professions — builders, farmers, teachers — each contributing unique functions.
When richness declines, specialization disappears; fewer species are left to perform critical biochemical tasks such as fiber fermentation, vitamin synthesis, or bile acid transformation.
Low richness is one of the earliest indicators of antibiotic-induced depletion or chronic GI stress, and recovery of richness is a key marker of microbiome restoration.
Total Fermenters —
The Energy Producers
The total fermenters index quantifies the abundance of bacteria responsible for producing short-chain fatty acids (SCFAs) such as acetate, propionate, and butyrate — the microbial “currency” of gut health.
These species (mainly Faecalibacterium, Blautia, Ruminococcus, and Lachnospiraceae) act as the city’s power plants, converting dietary fiber into energy for colonocytes and signaling molecules for the immune system. A decline in fermenters reflects functional exhaustion of the microbiome, often seen with chronic inflammation, malnutrition, or heavy antibiotic exposure. When fermenters recover, it’s a sign the ecosystem is rebuilding its metabolic strength and intestinal barrier support.
The Integrated Picture
By combining the DSI, tiered taxonomy, and these foundational indices, veterinarians gain a multidimensional view of the patient’s gut health:
- TBC shows the overall population size,
- Richness shows diversity,
- Evenness shows balance, and
- Total Fermenters show functional capacity.
Together, they transform microbiome testing from a descriptive list of microbes into a dynamic functional assessment — revealing not just what’s there, but how well it’s working and how severely it’s disrupted.
Toxins and the Microbiome
Invisible Disrupters to Gut Health
The gut microbiome is more than a collection of microbes — it’s a dynamic, symbiotic ecosystem that depends on a stable environment. When toxins enter the system, whether from food, water, soil, or the pet’s living space, they can directly or indirectly alter this microbial community, leading to dysbiosis, inflammation, and impaired metabolic function.

HEAVY METALS: Silent Saboteurs
Metals such as lead, mercury, aluminum, cadmium, and arsenic are among the most potent environmental stressors affecting microbial ecology.
- Mechanism: Heavy metals generate oxidative stress, damage microbial membranes, and interfere with bacterial enzymes involved in energy and nutrient metabolism. Beneficial anaerobes like Faecalibacterium and Ruminococcus are particularly sensitive, while resistant genera such as Enterococcus and Proteobacteria tend to proliferate.
- Functional Impact: Metal exposure reduces short-chain fatty acid (SCFA) production, impairs barrier integrity, and increases endotoxin translocation — creating a “leaky gut” phenotype.
- Clinical Relevance: Chronic low-level exposure in pets (via food, water, or toys) has been associated with persistent diarrhea, immune dysregulation, and loss of microbiome diversity.
Measuring and monitoring these metals alongside the Dysbiosis Severity Index (DSI) helps link environmental toxicity to microbiome health.
MYCOTOXINS: Biological Disruptors with Far-Reaching Effects
Aflatoxin, ochratoxin A, zearalenone, deoxynivalenol, trichothecenes, and gliotoxin — common fungal metabolites found in contaminated grains and pet foods — can dramatically alter intestinal microbiota.
- Mechanism: Mycotoxins damage the epithelial barrier and suppress beneficial SCFA-producing bacteria while promoting Proteobacteria and fungi.
- Functional Changes: Reduced butyrate and propionate levels compromise immune regulation, while mycotoxin-induced oxidative stress fosters opportunistic bacterial and yeast overgrowth.
- Clinical Presentation: Chronic exposure is linked to malabsorption, inflammatory enteropathies, and systemic immune or hepatic stress.
Because these toxins affect both microbial composition and host detoxification pathways, combining mycotoxin testing with DSI analysis provides a powerful integrative view of gut and metabolic health.
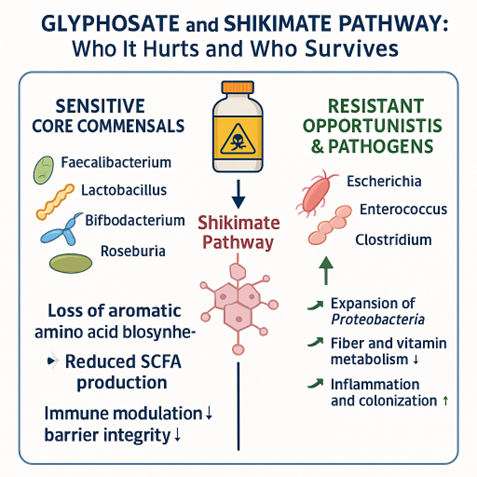
GLYPHOSATE: The Unintended Antibiotic
Glyphosate: The Unintended Antibiotic
Patented as a broad-spectrum antimicrobial, glyphosate inhibits the bacterial shikimate pathway — essential for synthesizing aromatic amino acids (tryptophan, tyrosine, phenylalanine).
- Mechanism: Many commensal bacteria (e.g., Lactobacillus, Bifidobacterium, Enterococcus) rely on this pathway and are sensitive to glyphosate exposure, while pathogens like Clostridium and Salmonella show higher resistance.
- Functional Impact: Loss of beneficial bacteria disrupts folate and vitamin B12 synthesis, reduces immune tolerance, and alters tryptophan-derived neurotransmitter balance along the gut–brain axis.
- Environmental Persistence: Pets encounter glyphosate through lawn treatments, contaminated grains, and groundwater, often at subclinical doses that accumulate over time.
| Category | Core Commensals Inhibited | Main Functional Loss |
| SCFA producers | Faecalibacterium, Roseburia, Blautia, Ruminococcus | Butyrate ↓, barrier integrity ↓ |
| Probiotics | Lactobacillus, Bifidobacterium | Immune modulation ↓, pathogen control ↓ |
| Carbohydrate fermenters | Prevotella, Eubacterium | Fiber metabolism ↓, gas and pH dysregulation |
| Mucin interactors | Akkermansia | Barrier regeneration ↓ |
| Vitamin synthesizers | Clostridium XIVa, Bacteroides | Folate/B12 synthesis ↓ |
Combined Burden: The Perfect Storm
Toxins rarely act in isolation. Heavy metals, mycotoxins, and glyphosate interact synergistically: metals enhance oxidative stress, mycotoxins weaken barrier function, and glyphosate suppresses microbial recovery.
The result is a self-reinforcing cycle of dysbiosis, inflammation, and impaired detoxification.
When these toxic exposures coincide with antibiotics or poor diet, the microbiome may lose resilience altogether, progressing toward chronic gastrointestinal disease, immune imbalance, and metabolic dysfunction.
Clinical Insight Through Integrated Testing
VDI’s comprehensive approach—linking Dysbiosis Severity Index (DSI) results with toxin and heavy-metal panels—allows veterinarians to pinpoint whether microbiome disruption stems from microbial imbalance alone or is driven by environmental toxicity.
This combined view transforms gut health evaluation from a local assessment to a whole-body ecosystem analysis, empowering clinicians to identify root causes and guide evidence-based detoxification, dietary, and microbiome-restoration strategies.
Toxin Detoxification
Restoring the Gut After Toxic Exposure
Every toxic exposure—whether from heavy metals, mycotoxins, or glyphosate—leaves a footprint on the microbiome. The Recovery Roadmap illustrates how the gut can heal when we remove the source, support detoxification, rebuild microbial balance, and monitor progress with objective data. Using VDI’s Dysbiosis Severity Index (DSI), veterinarians can track recovery step by step—seeing not only when the microbiome stabilizes, but how well it regains its protective, metabolic, and immune functions. Measure. Restore. Maintain.
Measure. Restore. Maintain.
VDI’s integrated toxin and microbiome testing platform gives veterinarians a complete picture of gut health—from disruption to full recovery.

| Toxin Type | Mechanism of Damage | Clinical Effect | Restoration Strategy |
| Heavy Metals (Lead, Mercury, Aluminum, Arsenic) | Generate oxidative stress and inhibit microbial enzymes; suppress SCFA producers like Faecalibacterium and Ruminococcus. | Loss of barrier integrity, diarrhea, immune dysregulation, and reduced microbiome diversity. | Identify exposure sources; use detox-supportive nutrition (fiber, antioxidants); monitor DSI and mineral status. |
| Mycotoxins (Aflatoxin, Ochratoxin A, DON, ZEN, Gliotoxin) | Damage intestinal epithelium and favor Proteobacteria and yeast expansion; reduce SCFA output. | Chronic GI inflammation, malabsorption, hepatic strain. | Remove contaminated feed, add mycotoxin binders and probiotic restoration; retest DSI for recovery. |
| Glyphosate | Inhibits bacterial shikimate pathway—impairs synthesis of aromatic amino acids in beneficial flora while sparing pathogens. | Reduced Lactobacillus and Bifidobacterium, altered vitamin B12/folate metabolism, neurotransmitter imbalance. | Minimize herbicide exposure (organic feed, filtered water); re-establish commensals with targeted probiotics. |
| Combined Burden | Metals amplify oxidative stress, mycotoxins weaken barrier, glyphosate hinders recovery—synergistic dysbiosis. | Persistent low-grade inflammation, immune dysfunction, chronic enteropathy. | Comprehensive detoxification plan; periodic toxin screening with DSI monitoring to gauge microbiome resilience. |
Restoring Balance After Toxic Exposure
When environmental toxins disrupt the gut, recovery depends on rebuilding both the microbial community and the host’s detoxification capacity. The goal isn’t just to remove toxins — it’s to re-establish a functional, resilient microbiome that protects the body from future insults.
1. Remove the Source
Identify and reduce ongoing exposure:
- Heavy metals: Filter drinking water, replace metal-contaminated dishes or toys, and use low-metal diets.
- Mycotoxins: Screen pet foods for contamination; avoid stored or mold-damaged kibble.
- Glyphosate: Choose foods made with organic or low-residue grains, and minimize lawn or garden herbicide use.
Source control is the foundation of recovery — no microbiome can heal in a toxic environment.
2. Support Detoxification
Provide nutrients and cofactors that aid hepatic and microbial clearance:
- Sulfur-rich amino acids (taurine, methionine) and antioxidants (vitamin E, selenium, glutathione precursors) enhance conjugation and reduce oxidative stress.
- Magnesium and zinc stabilize detox enzymes and cellular defense systems.
- Fiber (psyllium, beet pulp, inulin) binds metals and mycotoxins in the gut, promoting safe elimination.
3. Rebuild the Microbiome
Once exposure is limited and detox pathways supported, restore microbial function:
- Targeted probiotics and synbiotics—particularly Lactobacillus, Bifidobacterium, Faecalibacterium, and Clostridium XIVa strains—help re-seed core commensals.
- Prebiotic substrates such as arabinogalactan, FOS, and resistant starch feed fermenters and boost SCFA production.
- Diet diversity with soluble fiber, moderate fat, and high digestibility supports repopulation without excessive fermentation.
A recovering microbiome re-establishes the barrier, restores bile acid metabolism, and suppresses opportunistic overgrowth.
4. Monitor Recovery with the DSI
VDI’s Dysbiosis Severity Index (DSI) quantifies how effectively the gut community is regaining balance.
- A declining DSI score over time confirms successful detoxification and microbial repair.
- Persistent or worsening scores signal ongoing toxin load or incomplete microbial restoration.
Regular DSI testing transforms post-toxin management from guesswork to data-driven care.
5. Maintain Resilience
After recovery, periodic evaluation of toxin burden and DSI ensures the microbiome remains robust against future challenges. Combining preventive testing with sound nutrition and minimal unnecessary antibiotics helps keep the GI ecosystem stable, functional, and self-protecting — the foundation of whole-body health.
Vitamin D and B12
Hidden Drivers of Microbiome Health
The microbiome doesn’t function in isolation — it depends on a nutrient environment that supports both the host and its resident bacteria. Two of the most influential vitamins in this relationship are vitamin D and vitamin B12. Deficiencies in either are common in dogs and cats with chronic gastrointestinal disease and can directly alter the composition and function of the gut microbiota.
VDI’s Integrated GI Panels measure Vitamin D, Vitamin B12, and the Dysbiosis Severity Index (DSI) together — linking nutrient status with microbial function and disease risk.
“Measure the structure. Measure the power. Measure the balance.”
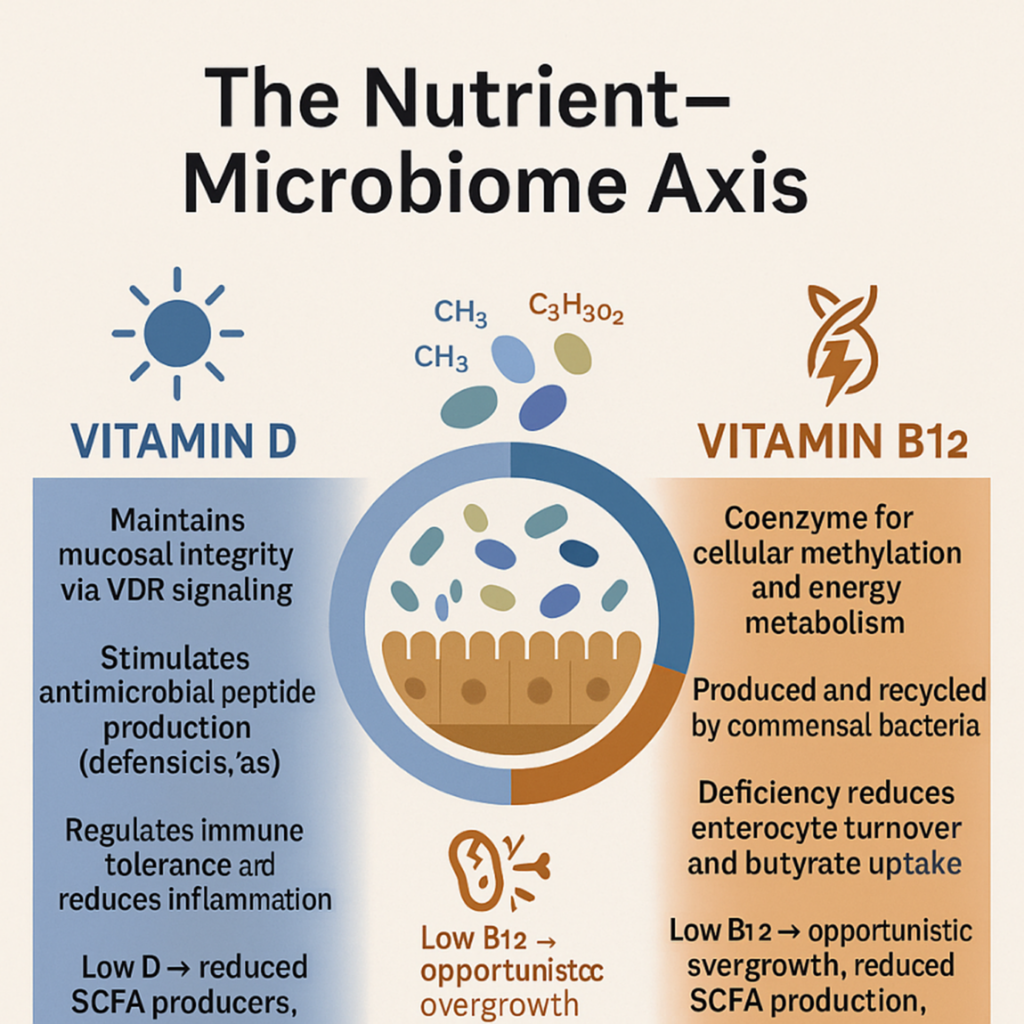
Vitamin D:
The Barrier Regulator
Vitamin D acts through the vitamin D receptor (VDR) expressed on intestinal epithelial cells and immune tissues throughout the gut.
- Microbiome Connection: VDR signaling regulates the production of antimicrobial peptides such as defensins and cathelicidins, maintaining epithelial integrity and controlling bacterial populations. Low vitamin D weakens this regulation, leading to overgrowth of opportunists (Proteobacteria, Enterococcus, Clostridium perfringens) and loss of core fermenters (Faecalibacterium, Blautia).
- Functional Impact: Deficiency reduces short-chain fatty acid production, compromises barrier tight junctions, and heightens mucosal inflammation.
Clinical Insight: In dogs and cats, low serum 25-hydroxy-vitamin D concentrations are frequently seen in chronic enteropathy, IBD, pancreatitis, and neoplasia — often reflecting both malabsorption and increased utilization.
Testing vitamin D provides a direct view into the mucosal immune tone and helps interpret whether dysbiosis is a cause or consequence of barrier failure
Vitamin B12 (Cobalamin):
The Metabolic Keystone
Vitamin B12 is central to DNA synthesis, methylation, and energy metabolism — but it’s also a microbiome-modulated nutrient.
- Microbiome Connection: Commensal bacteria synthesize and compete for B12; when dysbiosis develops, B12-producing species decline, and analog-producing or scavenging bacteria expand. This leads to functional deficiency even when serum B12 appears normal.
- Low B12 Effects: Deficiency slows enterocyte turnover, reduces butyrate uptake, and promotes anaerobic fermentation imbalances. Opportunists such as Bacteroides and Clostridium paraputificum may bloom under these low-B12 conditions, worsening dysbiosis.
- Clinical Insight: Hypocobalaminemia is strongly associated with chronic small intestinal disease in dogs and cats, exocrine pancreatic insufficiency, and malabsorptive syndromes. Restoring B12 improves not only metabolic parameters but also microbial balance and SCFA profiles.
When to Test Vitamin D & B12
- Chronic or recurrent gastrointestinal signs (vomiting, diarrhea, weight loss)
- Suspected malabsorption, IBD, or dysbiosis (moderate–high DSI)
- Long-term antibiotic, corticosteroid, or PPI therapy
- Chronic inflammation, pancreatitis, or cancer
- Unexplained fatigue, anorexia, or low muscle condition
Testing vitamin D and B12 alongside the Dysbiosis Severity Index (DSI) links metabolic capacity to microbial function — revealing whether the gut environment can sustain a healthy microbiome or if nutritional support is required to restore it.
Key Takeaway
Low vitamin D weakens the walls. Low B12 drains the power.
Together, they define how stable and functional the microbial community can be.
Monitoring these nutrients alongside the DSI provides veterinarians with a complete diagnostic view — connecting nutrition, immunity, and microbiome integrity in every GI patient.
Antibiotics Use
Use Wisely, Use When Needed
Antibiotics are invaluable when used appropriately — they save lives, resolve infections, and remain essential in veterinary medicine. But their power comes with risk. Each course disrupts the gut’s delicate microbial ecosystem, reducing diversity, suppressing beneficial fermenters, and allowing opportunistic or resistant bacteria to flourish. Repeated or unnecessary use can create long-lasting dysbiosis, impaired nutrient absorption, immune dysregulation, and increased susceptibility to chronic enteropathies.
Antibiotic therapy should be reserved for confirmed or strongly suspected bacterial infections supported by diagnostic evidence — such as positive culture, cytology, or systemic illness with fever and leukocytosis. In cases of acute, uncomplicated diarrhea, studies show that antibiotics rarely improve outcomes and may actually delay recovery. Supportive care, dietary management, probiotics, and microbiome monitoring are safer first-line strategies.
By prescribing thoughtfully and monitoring gut health with tools like the Dysbiosis Severity Index (DSI), veterinarians can protect both patient wellbeing and the long-term effectiveness of antibiotics — ensuring they remain a remedy, not a recurring cause of disease.

Responsible Antibiotic Use: Protecting the Microbiome
| Use Only When Indicated | Confirm or strongly suspect bacterial infection through culture, cytology, or systemic evidence. Avoid empirical use for mild or self-limiting GI signs. |
| Target Precisely | Choose narrow-spectrum antibiotics whenever possible. Adjust based on sensitivity results and discontinue when infection control is achieved. |
| Limit Exposure | Use the shortest effective duration; extended courses amplify dysbiosis and resistance. Avoid “just in case” prescriptions that offer little therapeutic benefit. |
| Support Recovery | Pair treatment with probiotics, synbiotics, and dietary support to help restore microbial balance. Re-evaluate gut health post-therapy with VDI’s Dysbiosis Severity Index (DSI) to monitor recovery and guide next steps. |
| Think Long-Term | Every antibiotic course reshapes the microbiome. Stewardship today preserves both patient health and the effectiveness of antimicrobial therapy tomorrow. |

