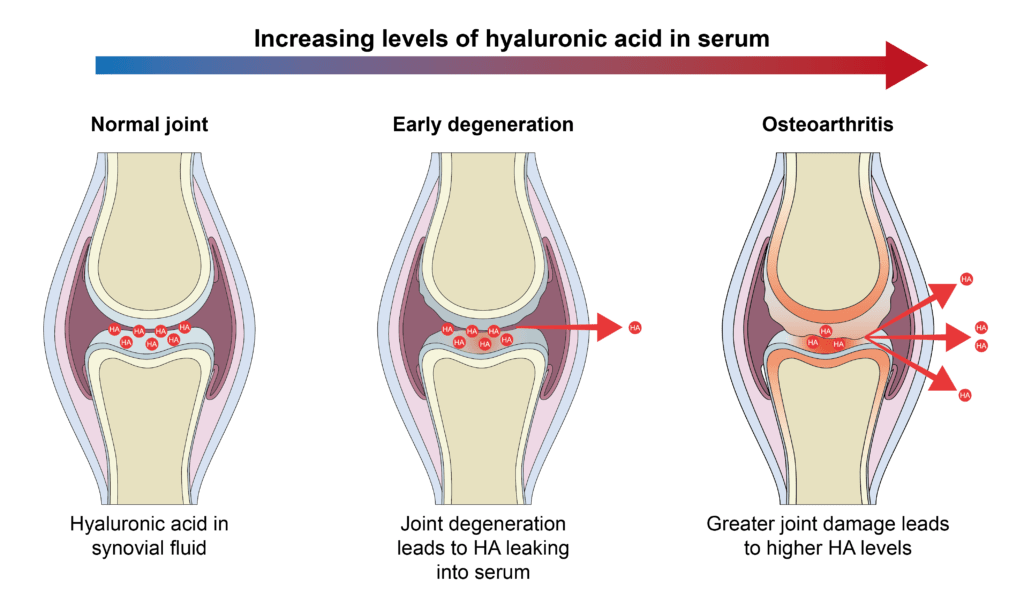
There is a growing body of evidence that Hyaluronic Acid is an effective marker of degenerative joint diseases in dogs:
Background:
Serum hyaluronic acid (sHA) and CRP are a simple blood test for the early detection of degenerative joint disease as well as post-therapeutic monitoring for therapy effectiveness. Positive sHA values are to be interpreted as positive for DJD provided the patient does not have severe liver disease (see Liver Disease). The use of CRP assists in the severity of osteoarthritis, treatment efficacy, and the differential diagnosis of IMPA. Patients using HA containing supplements should be taken off supplements 48 hours prior to blood draw to prevent interference. Screening for OA should be in dogs 5 years of age and older and routinely in high-risk breeds.
Table of Contents:
- The Synovial Joint
- Degenerative Joint Disease
- Hyaluronic Acid – a DJD Biomarker
- Inflammation in DJD
- OA Treatment
- Hyaluronan – Injection and Oral
- Liver Disease
- References
The Synovial Joint
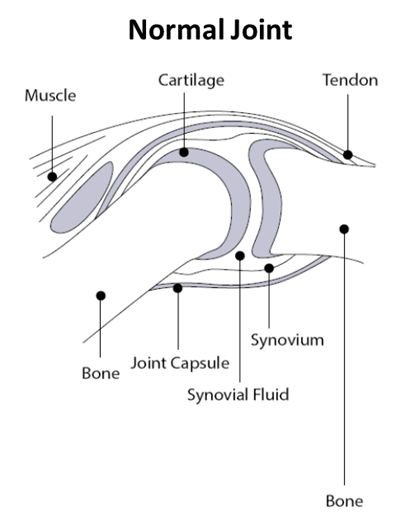
Synovial fluid, found in the cavities of synovial joints, provides lubrication to reduce friction during movement. Synovial fluid is an ultra-filtrate of blood plasma; its composition regulated by the synovial membrane. Within the joint are chondrocytes which are responsible for the extracellular matrix composition (ECM), regulation, and homeostasis. Normal joint cartilage consists of 5% chondrocytes and 95% ECM (1).
A key component of synovial fluid is hyaluronan, also known as hyaluronic acid (HA). HA, a glycosaminoglycan, is found in the skin, synovial fluid and other tissues, and has a unique capacity to bind and retain water molecules achieving excellent viscoelasticity. These properties make HA act as a lubricant and shock absorber, extremely useful in joints (1). Chondrocytes, cells within the articular cartridge, are responsible for the synthesis of synovial fluid including HA (2).
Degenerative Joint Disease
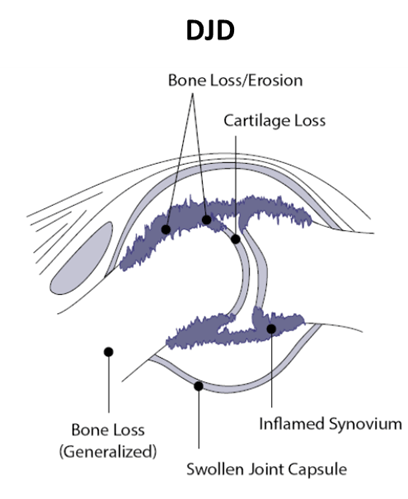
Degenerative joint disease (DJD) is a common, age-related breakdown of the joint. Other risk factors include weight, breed, genetic line, and diet. Osteoarthritis (OA) is the most commonly diagnosed DJD in both human and veterinary medicine and affects 20% of dogs older than one year and a stunning 80% in dogs greater than 8 years (3). Certain breeds are predisposed to OA with males having a higher incidence than females, especially in neutered dogs (4)(5). Other DJD include intervertebral disk disease (IVDD), degenerative myelopathy (DM), and immune mediated polyarthritis (IMPA). Sooner or later, many dogs succumb to the crippling effects of DJD with most not being clinically diagnosed until it reaches an advanced stage and clinical signs are obvious (6).
DJD is characterized by changes in the ECM, including bone loss, inflammation, and oxidative stress. Increases in IL-1, TNF-α, and other pro-inflammatory cytokines occur. The synovial membrane, which regulates the synovial fluid composition, develops synovitis. There appears to be two major causes of synovitis – age related and immune mediated. OA is typically a “wear ‘n tear” age related disease in contrast to IMPA which is primarily an autoimmune disease.
Loss of synovial fluid into the peripheral blood due to synovitis induces joint friction, more inflammation, pain, and stiffness. There appears to be a correlation between the degree of cartilage damage and chondrocyte death (7). Over time, chondrocyte death and ECM loss form a vicious cycle with the progression of one aggravating the other. The exact mechanism of DJD initiation and propagation is not well understood, however a key driver is dysregulation of the innate immune response and production of pro-inflammatory cytokines (8).
Hyaluronic Acid – A DJD Biomarker
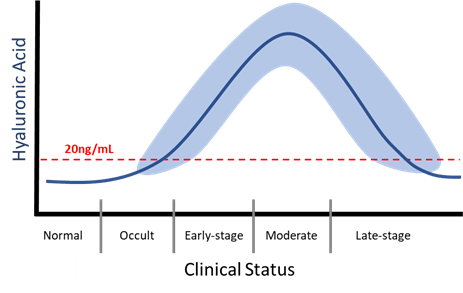
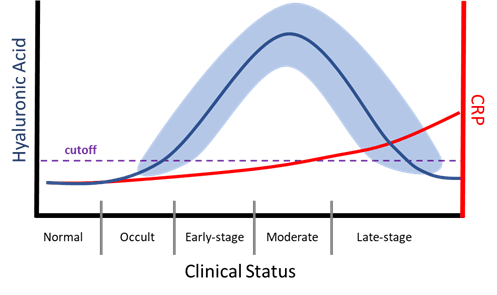
The use of HA has been examined by a number of investigators in both human and veterinary medicine. The concept is simple: synovitis due to DJD allows HA to leak into the peripheral blood. HA has a very short half-life of 5 minutes therefore elevated serum HA (sHA) values correlate to the onset of DJD.
In the early to moderate stages of OA, serum concentration of HA (sHA) increases as the chondrocytes continue to replace HA leaking out into the blood stream (7)(8). Increasing concentration of sHA correlates with disease severity (9). As disease worsens there is a decline in sHA as chondrocytes have diminished capacity to produce HA, there is an increase in proteolytic enzymes destroying HA, and ultimately chondrocyte death in late-state disease. Depending upon treatment, sHA level can increase again (see OA Treatment).
Inflammation in DJD
DJD ultimately is an inflammatory disease however the initiation of disease could be an injury or poor conformation. Most of the inflammation is localized at the joint and typically does not produce measurable levels of serum CRP until later; only IMPA will produce significant CRP due to its autoimmune nature. In OA, inflammation, and serum CRP, will progressively get worse as mechanical damage, bone loss, and chondrocyte death occurs. In an untreated patient, CRP will continually increase. With treatment, inflammation should be controlled and return to normal.
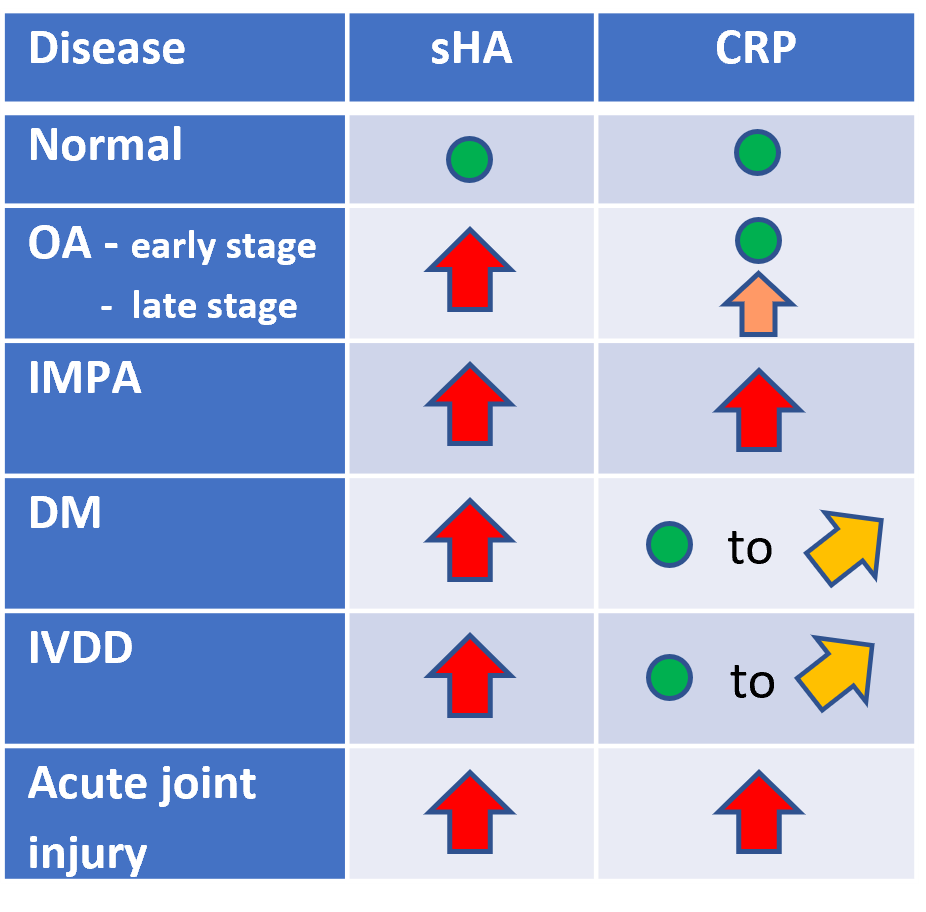
IMPA significantly differs from other OA in its autoimmune origin. It is classified based upon whether it is erosive or non-erosive. IMPA is believed to be a result of immune-complex deposition within the synovium resulting in a sterile synovitis. The antigenic stimuli can be from a wide range or sources such as a distant infection, neoplasia, drug reaction, and others. This antigenic stimulation of the immune system triggers a moderate to strong inflammatory response resulting in elevated concentrations of CRP at disease onset. Treatment for IMPA is immunosuppressive doses of prednisone and prognosis is favorable especially when caught early (10)(11).
DM presents primarily in older dogs with German Shepherds being the most common. DM is necrosis within the spinal cord segments with secondary demyelination. DM presents as a slowly progressive, non-painful myelopathy that will over time progress cranially (12). There is no known cure for DM. DM will present with elevated sHA and normal to mildly elevated CRP.
IVDD is clinically similar to DM and the differential diagnosis can be difficult. Both result in degeneration of the spinal cord. IVDD is believed to be a reduction of nutrients to the spinal cord causing chondrocyte death and changes in the ECM (13). There is no known cure for IVDD. IVDD will present with elevated sHA and normal to mildly elevated CRP.
OA Treatment
NSAIDS
NSAIDS are used to control inflammation and to provide patient comfort. When used early and consistently studies show disease progression is slowed and overall quality of life is increased. NSAIDS do not stimulate the production of hyaluronic acid. Both short and long-term acting COX-2 inhibitors (eg, Carprofen, Mavacoxib) perform similarly (14).
Adequan®
Adequan® or polysulfated glycosaminoglycan (PSGAG) is an intramuscular injection for the control of signs associated with non-infectious degenerative and/or traumatic arthritis of canine synovial joints. While the exact mechanism of action is unknown, PSGAG is known to inhibit proteolytic enzymes which includes hyaluronidases (15). VDI’s own internal data shows that patients treated with PSGAG have increasing concentrations of sHA. In a study looking at both PSGAG and intra-articularly injected hyaluronan, both preformed equally in improving mobility (16).
Adequan is a registered trademark of America Regent, Inc.
Stem Cells/ PRP
While NSAIDS remain the mainstay therapy, other treatment modalities such as mesenchymal stem cell (MSC) therapy or platelet rich plasma (PRP) are becoming more common when NSAIDS fail to provide the relief needed.
MSC therapy is focused on their anti-inflammatory, immunomodulatory, and immunosuppressive potential. Stem cells can be harvested from a variety of sources however the most used is adipose tissue. While the exact mechanism of action is still being investigated, in-vitro studies show stem cells can differentiate toward chondrocytes (17). With successful treatment the concentration of sHA is increased (7).
PRP is rich in platelet-derived growth factors. The logic behind PRP is a supra-physiological amount of growth factors delivered directly at the site of the damaged joint. The goal is to promote natural healing and tissue regeneration (18). Studies mostly focus on its ability to heal fractures. Use in OA has limited data however in one study use of PRGR did increase sHA concentration, indicating a stimulating effect on the chondrocytes (7).
Hyaluronan – Injection and Oral
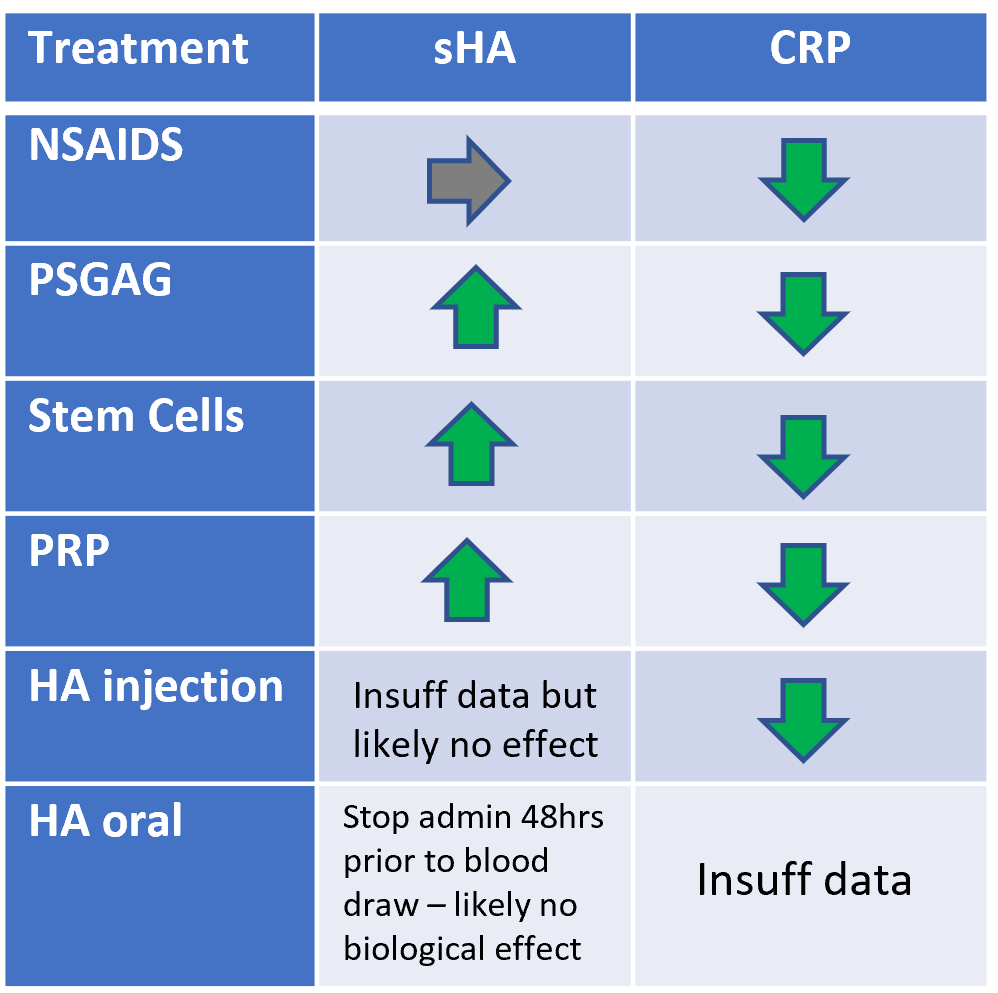
Intra-articular injection of HA (IA-HA) is an established procedure in both human and veterinary medicine. IA-HA improves shock absorption, joint lubrication, anti-inflammatory effects, chondroprotection, and improvements to the ECM. Canine studies show improvement in lameness within 1week and for up to 6 months post injection. Combined therapy with corticosteroids shows additional benefit although the use of corticosteroids long term has significant disadvantages (19).
Supplements containing oral hyaluronan are widely available. Whereas IA-HA uses high molecular weight HA, oral HA containing supplements use low molecular weight HA. Studies in both human and veterinary medicine varies in oral HA efficacy, however the theory is the same as in IA-HA – to reduce inflammation and to be absorbed by the joints. A major problem with oral HA is how fast the liver degrades HA reducing the amount of HA to be delivered to its target. Canine studies show some improvement after prolonged use (20).
Liver Disease
HA is widely present in the body’s ECM. The liver rapidly destroys circulating HA; the half-life of HA is less than 5 minutes. In severe liver disease, the destruction of HA is delayed resulting in higher sHA values. Dogs with severe liver disease may present with high levels of sHA and not have DJD.
The type of liver disease that will increase sHA is limited to chronic hepatitis and chronic cirrhosis that produces liver fibrosis as well as dogs with congenital portosystemic shunt. In these patients, hepatic fibrosis in response to an acute or chronic liver injury to parenchymal cells curtails the degradation of HA raising sHA levels. In situations where performing a liver biopsy is not possible, studies show sHA level to be a reliable indicator of hepatic fibrosis with a sensitivity of 82% and specificity of 89% (21)(22).
References
- Gupta RC, Lall R, Srivastava A, Sinha A. Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory. Front Vet Sci. 2019;6:192. Published 2019 Jun 25. doi:10.3389/fvets.2019.00192
- Akkiraju H, Nohe A. Role of Chondrocytes in Cartilage Formation, Progression of Osteoarthritis and Cartilage Regeneration. J Dev Biol. 2015;3(4):177-192. doi:10.3390/jdb3040177
- Anderson KL, O’Neill DG, Brodbelt DC, et al. Prevalence, duration and risk factors for appendicular osteoarthritis in a UK dog population under primary veterinary care. Sci Rep. 2018;8(1):5641. Published 2018 Apr 4. doi:10.1038/s41598-018-23940-z
- Hays L, Zhang Z, Mateescu RG, Lust G, Burton-Wurster NI, Todhunter RJ. Quantitative genetics of secondary hip joint osteoarthritis in a Labrador Retriever-Greyhound pedigree. Am J Vet Res. 2007;68(1):35-41. doi:10.2460/ajvr.68.1.35
- Hart BL, Hart LA, Thigpen AP, Willits NH. Long-term health effects of neutering dogs: comparison of Labrador Retrievers with Golden Retrievers. PLoS One. 2014;9(7):e102241. Published 2014 Jul 14. doi:10.1371/journal.pone.0102241
- Pettitt RA, German AJ. Investigation and management of canine osteoarthritis. In Practice. 2015;37:1–8. doi: 10.1136/inp.h5763.
- Vilar JM, Rubio M, Spinella G, Cuervo B, Sopena J, Cugat R, Garcia-Balletbó M, Dominguez JM, Granados M, Tvarijonaviciute A, Ceron JJ, Carrillo JM. Serum Collagen Type II Cleavage Epitope and Serum Hyaluronic Acid as Biomarkers for Treatment Monitoring of Dogs with Hip Osteoarthritis. PLoS One. 2016 Feb 17;11(2):e0149472. doi: 10.1371/journal.pone.0149472.
- Mathiessen A, Conaghan PG. Synovitis in osteoarthritis: current understanding with therapeutic implications. Arthritis Res Ther. 2017;19(1):18. Published 2017 Feb 2. doi:10.1186/s13075-017-1229-9.
- Sasaki E, Tsuda E, Yamamoto Y, et al. Serum hyaluronic acid concentration predicts the progression of joint space narrowing in normal knees and established knee osteoarthritis – a five-year prospective cohort study. Arthritis Res Ther. 2015;17:283. Published 2015 Oct 10.
- Wilson-Wamboldt J. Type I idiopathic non-erosive immune-mediated polyarthritis in a mixed-breed dog. Can Vet J. 2011;52(2):192-196.
- Foster JD, Sample S, Kohler R, Watson K, Muir P, Trepanier LA. Serum biomarkers of clinical and cytologic response in dogs with idiopathic immune-mediated polyarthropathy. J Vet Intern Med. 2014 May-Jun;28(3):905-11. doi: 10.1111/jvim.12351.
- Story BD, Miller ME, Bradbury AM, et al. Canine Models of Inherited Musculoskeletal and Neurodegenerative Diseases. Front Vet Sci. 2020;7:80. Published 2020 Mar 11. doi:10.3389/fvets.2020.00080.
- Jeffery ND, Levine JM, Olby NJ, Stein VM. Intervertebral disk degeneration in dogs: consequences, diagnosis, treatment, and future directions. J Vet Intern Med. 2013 Nov-Dec;27(6):1318-33. doi: 10.1111/jvim.12183.
- Payne-Johnson M, Becskei C, Chaudhry Y, Stegemann MR. Comparative efficacy and safety of mavacoxib and carprofen in the treatment of canine osteoarthritis. Vet Rec. 2015 Mar 14;176(11):284. doi: 10.1136/vr.102397.
- Adequan Canine package insert.
- Frisbie DD, Kawcak CE, McIlwraith CW, Werpy NM. Evaluation of polysulfated glycosaminoglycan or sodium hyaluronan administered intra-articularly for treatment of horses with experimentally induced osteoarthritis. Am J Vet Res. 2009 Feb;70(2):203-9. doi: 10.2460/ajvr.70.2.203.
- Dias IE, Cardoso DF, Soares CS, et al. Clinical application of mesenchymal stem cells therapy in musculoskeletal injuries in dogs-a review of the scientific literature. Open Vet J. 2021;11(2):188-202. doi:10.5455/OVJ.2021.v11.i2.2.
- Raeissadat SA, Gharooee Ahangar A, Rayegani SM, Minator Sajjadi M, Ebrahimpour A, Yavari P. Platelet-Rich Plasma-Derived Growth Factor vs Hyaluronic Acid Injection in the Individuals with Knee Osteoarthritis: A One Year Randomized Clinical Trial. J Pain Res. 2020 Jul 8;13:1699-1711. doi: 10.2147/JPR.S210715.
- Franklin SP, Cook JL. Prospective trial of autologous conditioned plasma versus hyaluronan plus corticosteroid for elbow osteoarthritis in dogs. Can Vet J. 2013;54(9):881-884.
- Martí-Angulo S, García-López N, Díaz-Ramos A. Efficacy of an oral hyaluronate and collagen supplement as a preventive treatment of elbow dysplasia. J Vet Sci. 2014;15(4):569-574. doi:10.4142/jvs.2014.15.4.569
- Ceplecha V, Řeháková K, Lendon C, Proks P, Miša Škorič M, Grinwis G, Hřibová B, Vávra M, Lorenzová J, Crha M. Hyaluronic acid and TGF-β1 in dogs with hepatobiliary diseases. Acta Vet. Brno 2018, 87: 231-240 https://doi.org/10.2754/avb201887030231.
- Eulenberg VM, Lidbury JA. Hepatic Fibrosis in Dogs. J Vet Intern Med. 2018 Jan;32(1):26-41. doi: 10.1111/jvim.14891. Epub 2017 Nov 30. PMID: 29194760.